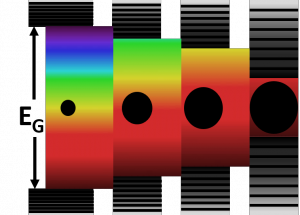
 Efforts are being made to design organic and inorganic hybrid structures that exhibit improved material functionality toward emissive materials, photovoltaic, sensing, data storage, nano-electronics and stimuli-responsive materials. Of particular interest are the nanocrystals as these materials offer a good strategy to construct low cost high performance optoelectronics and smart materials in the future. One of the fundamental properties of a semiconductor is the presence of a band gap separating the occupied valence band from the empty conduction band states. The band gap is defined by the materials identity for a bulk semiconductor whereas it is controlled by the size for quantum dots. As the size of a particle approaches to/or less than a size which is smaller than its Bohr radius, the electron and hole interactions with the particle surface will dominate the dynamics.
Efforts are being made to design organic and inorganic hybrid structures that exhibit improved material functionality toward emissive materials, photovoltaic, sensing, data storage, nano-electronics and stimuli-responsive materials. Of particular interest are the nanocrystals as these materials offer a good strategy to construct low cost high performance optoelectronics and smart materials in the future. One of the fundamental properties of a semiconductor is the presence of a band gap separating the occupied valence band from the empty conduction band states. The band gap is defined by the materials identity for a bulk semiconductor whereas it is controlled by the size for quantum dots. As the size of a particle approaches to/or less than a size which is smaller than its Bohr radius, the electron and hole interactions with the particle surface will dominate the dynamics.
Decreasing the size of particles to less than their Bohr radius results in confinement of electron and hole wavefunctions. In this size range, the band gap is controlled by the particle size in three-dimension and this phenomenon is called as the “quantum size effect”. The nanoscale particles which exhibit this property are referred to as quantum dots (QDs). The most remarkable property of these materials is the massive changes in optical properties as a function of the size. As the size is reduced, the
electronic transitions shift to the higher energy.
Semiconductor structures can only operate at their potential only if they can be grown with a high degree of crystallinity. Controlling defects and impurities is important for high structural quality. In our group, we are trying to find synthetic and postsynthsis methods to control the surface states of nanocrystals.
http://onlinelibrary.wiley.com/doi/10.1002/adfm.201401816/full
References:
Brus, L. E. Electron–electron and electron-hole interactions in small semiconductor crystallites: The size dependence of the lowest excited electronic state. J. Chem. Phys. 80, 4403 (1984).
Klimov, V. Nanocrystal Quantum Dots, Second Edition. (CRC Press, 2010).
Alivisatos, A. P. Perspectives on the Physical Chemistry of Semiconductor Nanocrystals. J. Phys. Chem. 100, 13226–13239 (1996).
